|
For neurodegenerative disorders associated with protein aggregation, the rationale has been that inhibition of Hsp90 activates heat shock factor-1 (HSF-1) to induce production of Hsp70 and Hsp40, as well as of other chaperones, which in turn, promote disaggregation and protein degradation. Hsp90 is a molecular chaperone with important roles in maintaining the functional stability and viability of cells under a transforming pressure. This mini-review will summarize our current knowledge on Hsp90 in neurodegeneration and will focus on the potential beneficial application of Hsp90 inhibitors in neurodegenerative diseases. Second, by activation of HSF-1 and the subsequent induction of heat shock proteins, such as Hsp70, Hsp90 inhibitors may redirect neuronal aggregate formation, and protect against protein toxicity. First, by suppressing aberrant neuronal activity, Hsp90 inhibitors may ameliorate protein aggregation and its associated toxicity. These biological functions therefore propose Hsp90 inhibition as a dual therapeutic modality in neurodegenerative diseases. In addition, Hsp90 regulates the activity of the transcription factor heat shock factor-1 (HSF-1), the master regulator of the heat shock response, mechanism that cells use for protection when exposed to conditions of stress. 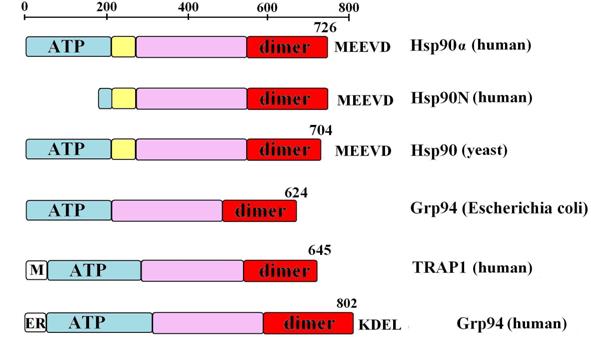
In addition to its well-characterized functions in malignancy, recent evidence from several laboratories suggests a role for Hsp90 in maintaining the functional stability of neuronal proteins of aberrant capacity, whether mutated or over-activated, allowing and sustaining the accumulation of toxic aggregates. Hsp90 is a molecular chaperone with important roles in regulating pathogenic transformation.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
